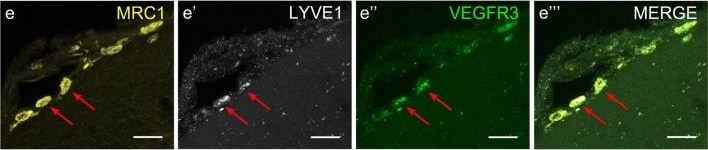
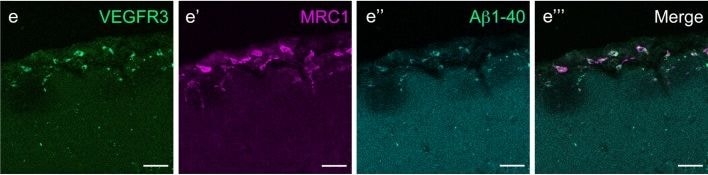
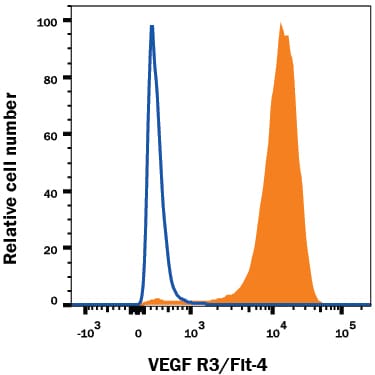
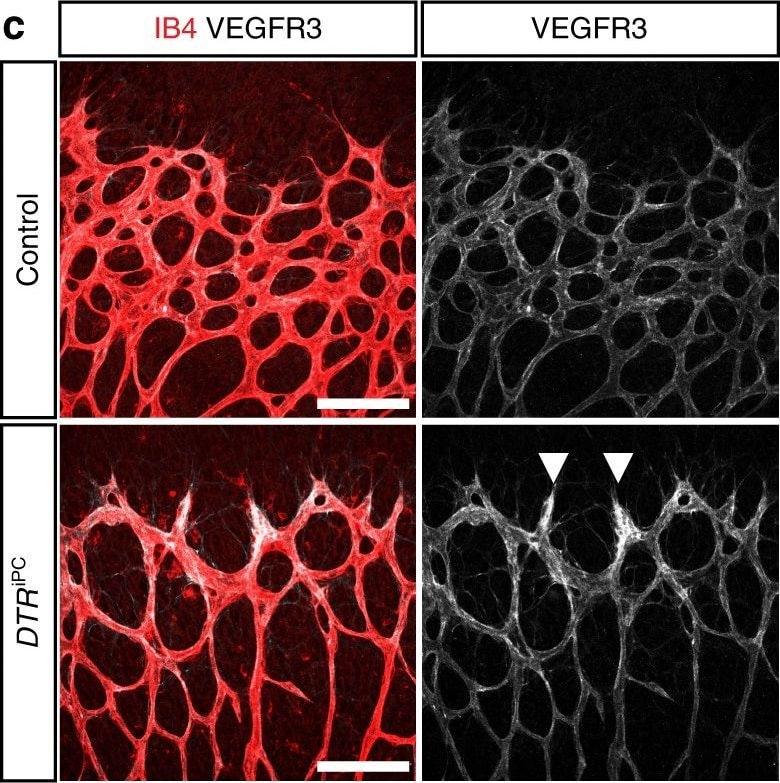
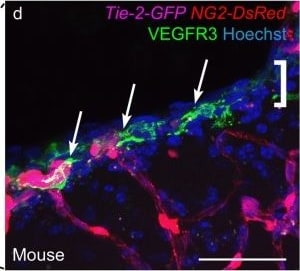
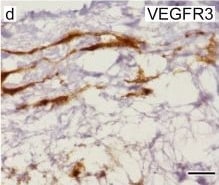
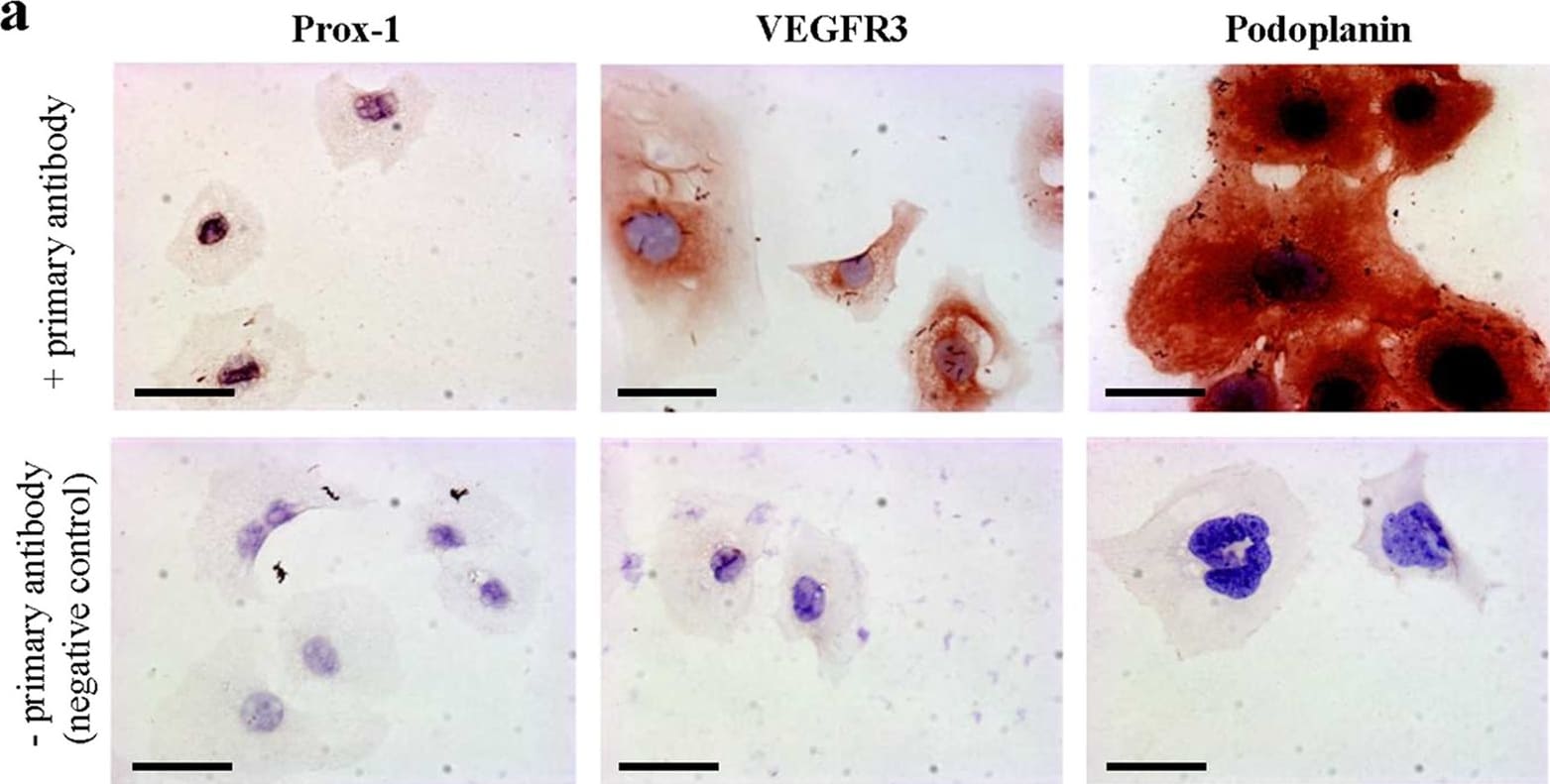
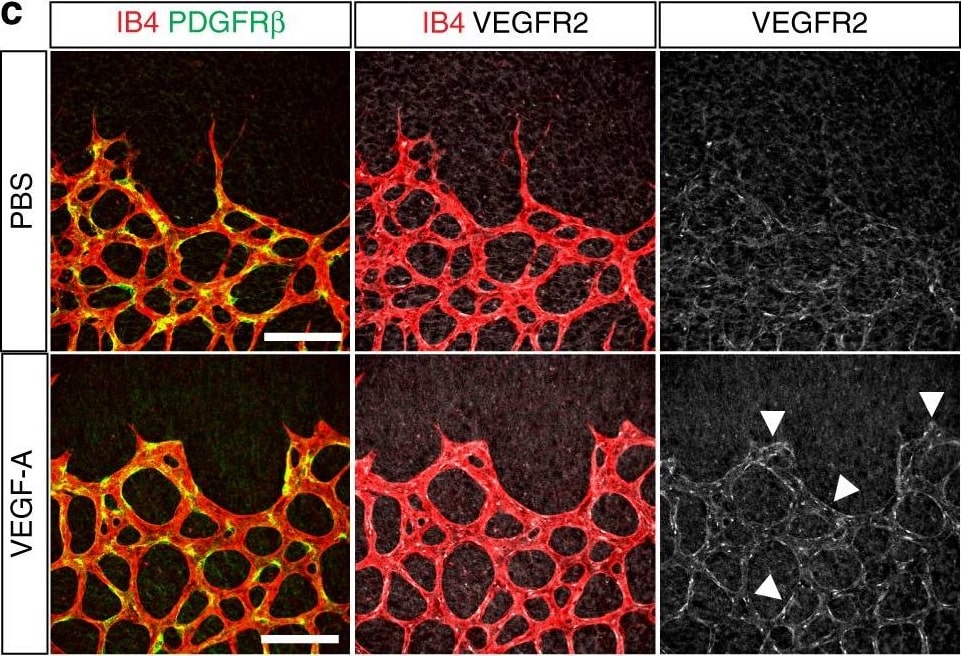
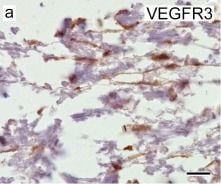
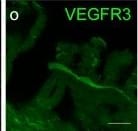
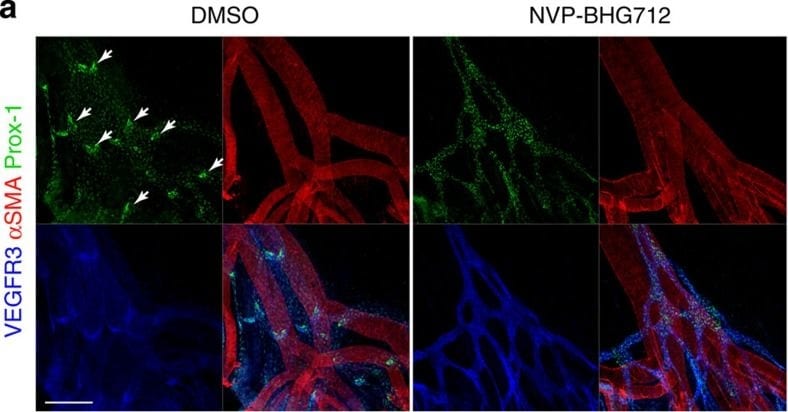
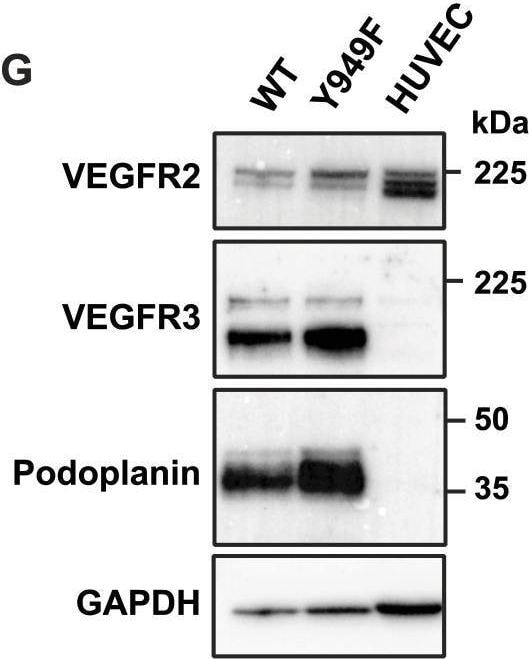
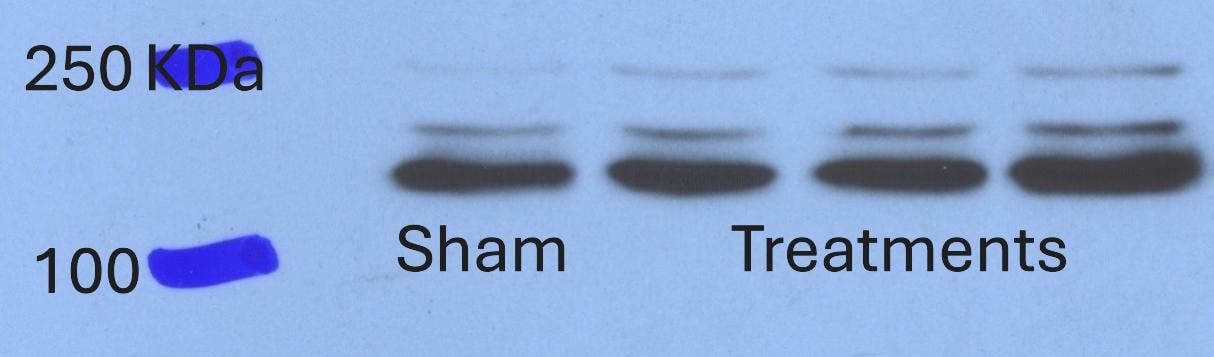
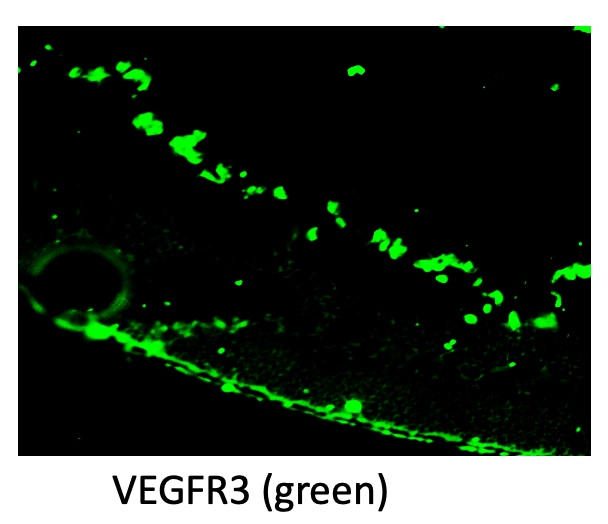
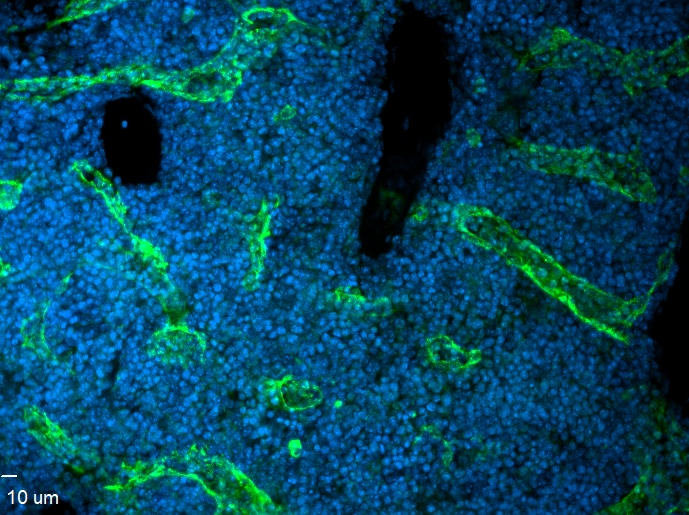
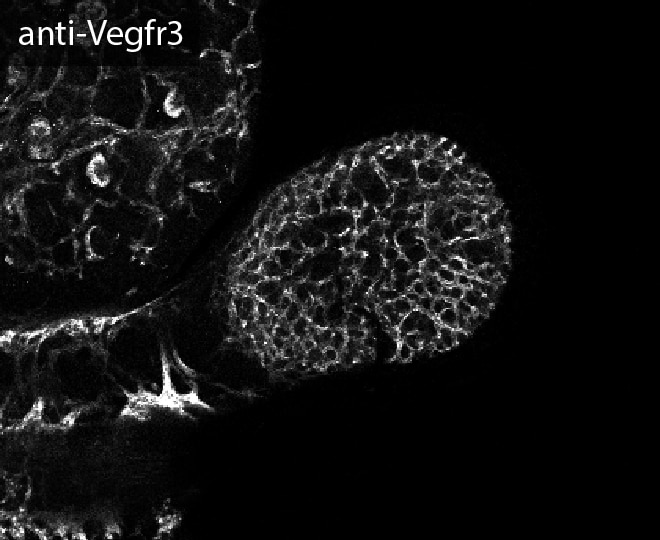
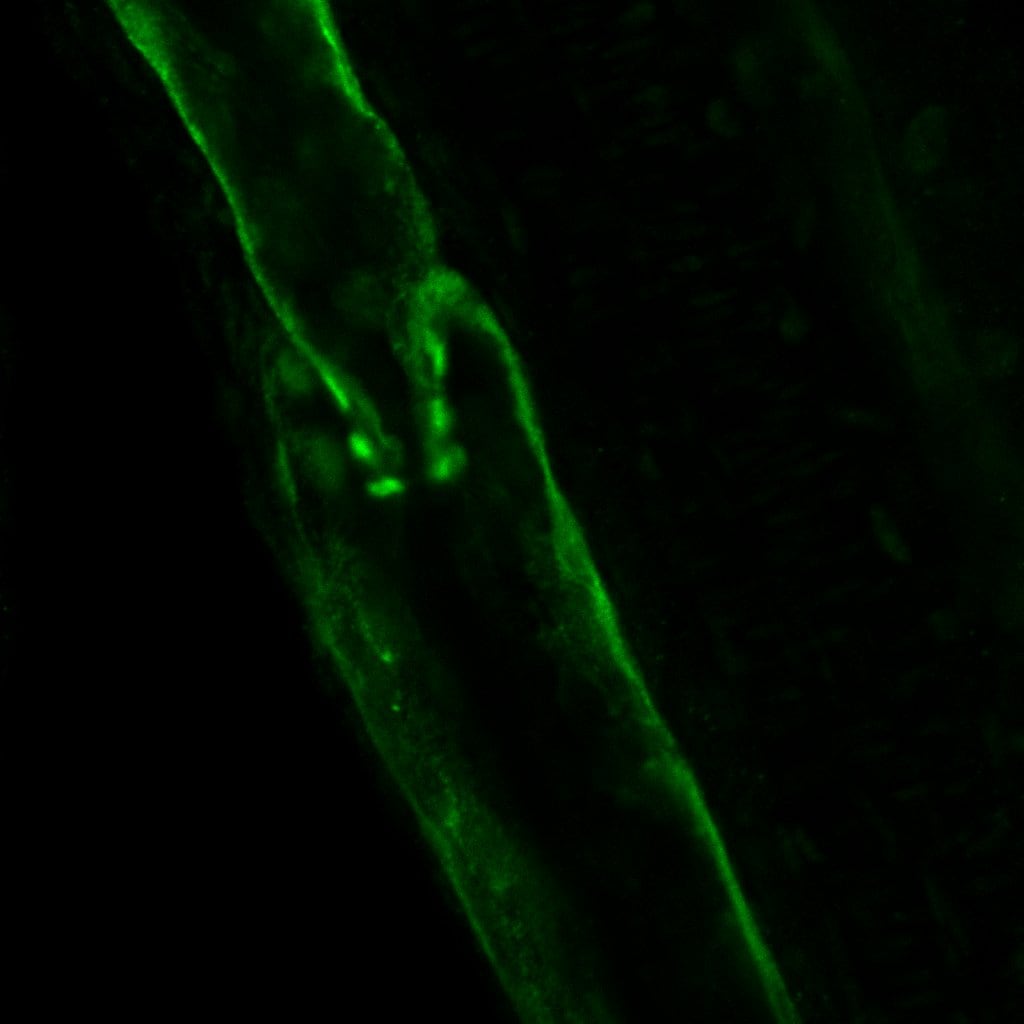
VEGFR3 (Flt-4), together with VEGFR1 (Flt-1) and VEGFR2 (KDR/Flk-1), belong to the class III subfamily of receptor tyrosine kinases (RTKs). All three receptors contain seven immunoglobulin-like repeats in their extracellular domains and kinase insert domains in their intracellular regions. The expression of these receptors is almost exclusively restricted to the endothelial cells. These receptors are likely to play essential roles in vasculogenesis and angiogenesis.
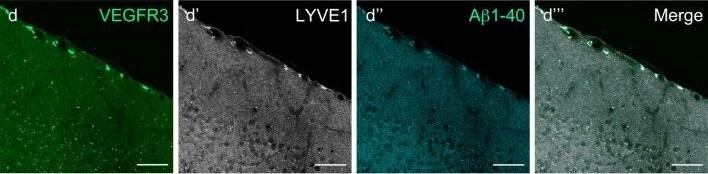
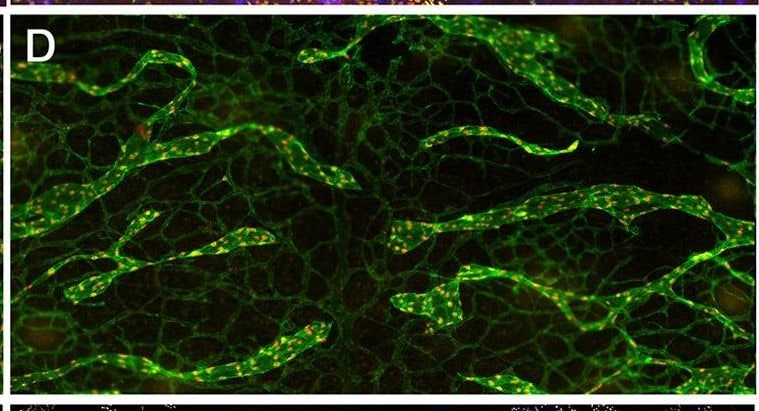
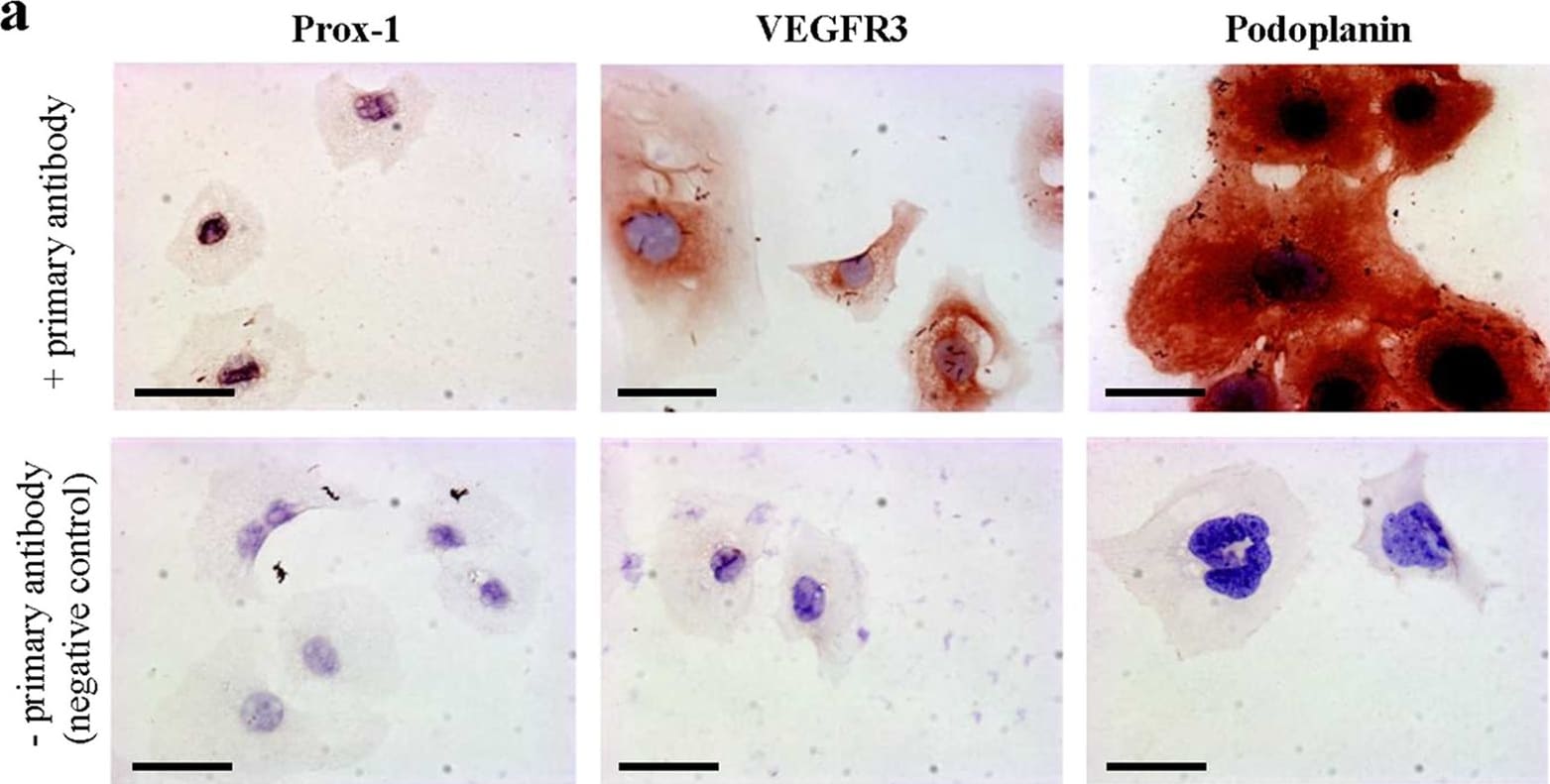
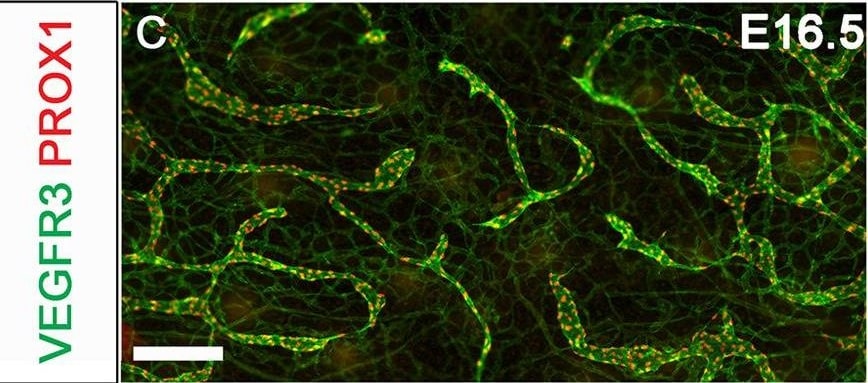
In adults, VEGFR3 expression is restricted to the endothelial cells of the lymphatic vessels. Mouse VEGFR3 cDNA encodes a 1363 amino acid (aa) residue precursor protein with a 24 aa residue signal peptide. Mature VEGFR3 has a 751 aa residue extracellular domain, a 22 aa residue hydrophobic transmembrane domain and a 565 aa residue cytoplasmic domain. The polypeptide sequences of murine Flt-4 is 88% identical to the human homologue. VEGFR3 has been reported to serve as the receptors for VEGF-C and VEGF-D.